Welcome to the Byers Group!

A central theme to research in our laboratory is the investigation of novel or underdeveloped organotransition metal chemistry and its catalytic application to useful processes in the fields of organic chemistry, inorganic chemistry, and materials science.
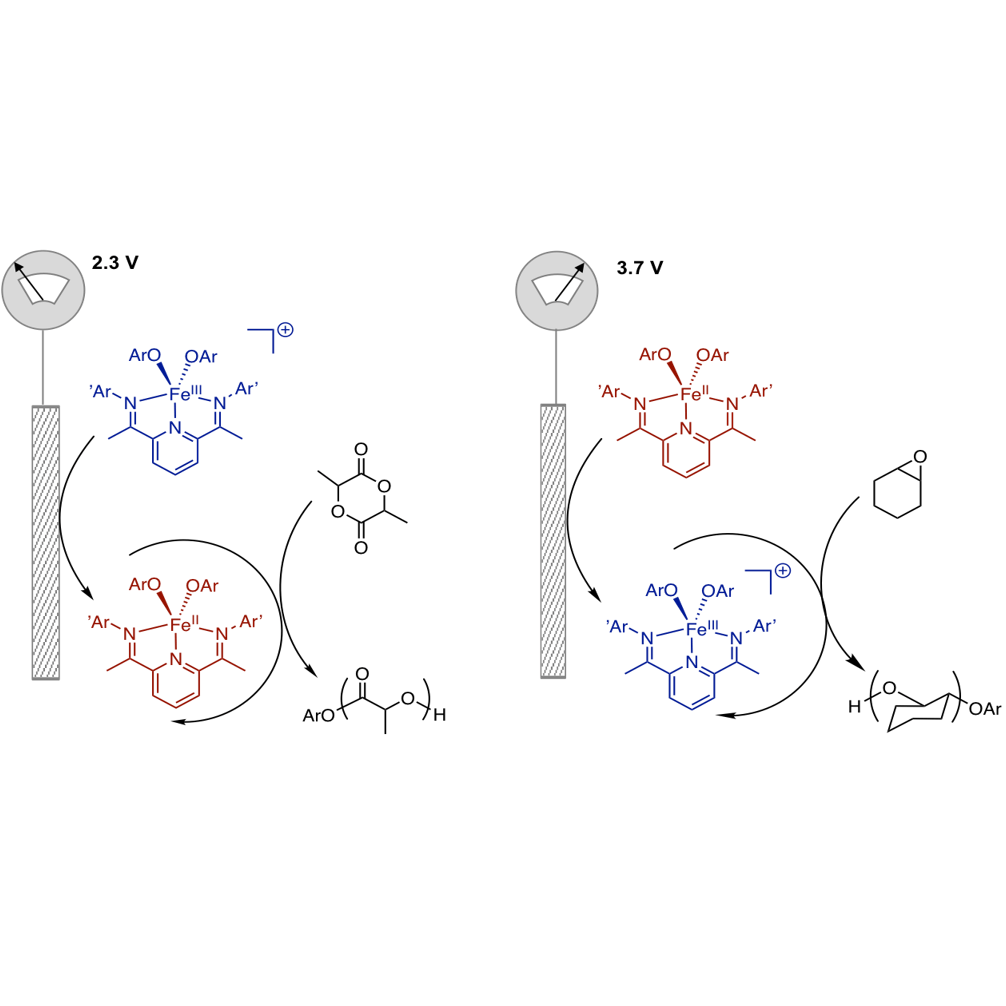
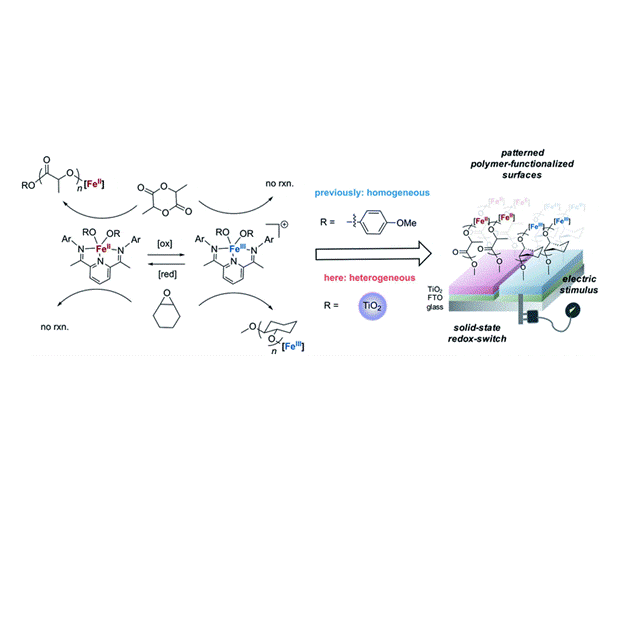
One of our major areas of interest is the development of new synthetic techniques for the production of novel copolymers useful as traditional engineering polymers, biodegradable polymers, or polymers useful for biomedical applications. To access these new materials, we have been developing iron catalysts that have tunable Lewis acidic properties as well as available one- and two-electron redox pathways to control composition, stereoselectivity, and architecture.
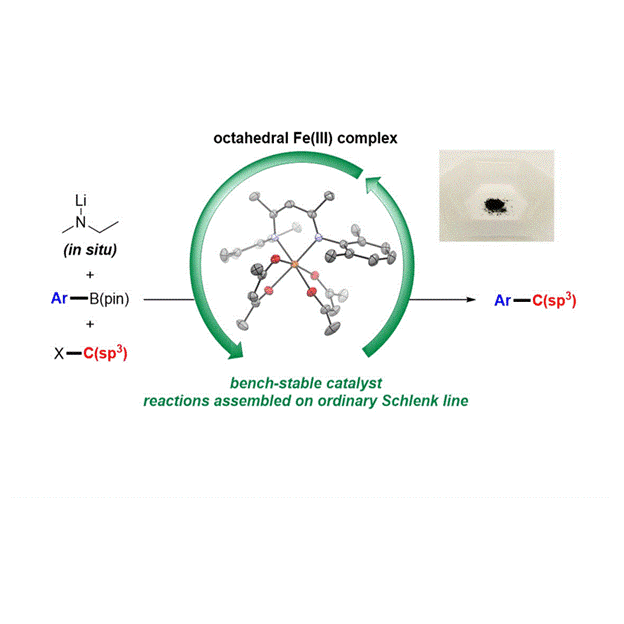
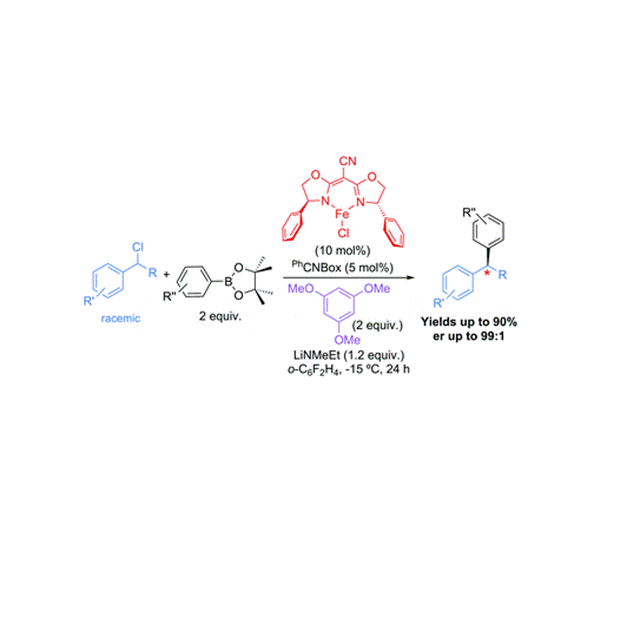
Another area of interest is in the development of cross coupling reactions involving earth abundant transition metal catalysts that are less expensive and toxic than state-of-the-art catalysts. In addition to the environmental advantages of these catalysts, the unique reactivity of these complexes makes them amenable towards expanding the scope of cross coupling reactions.
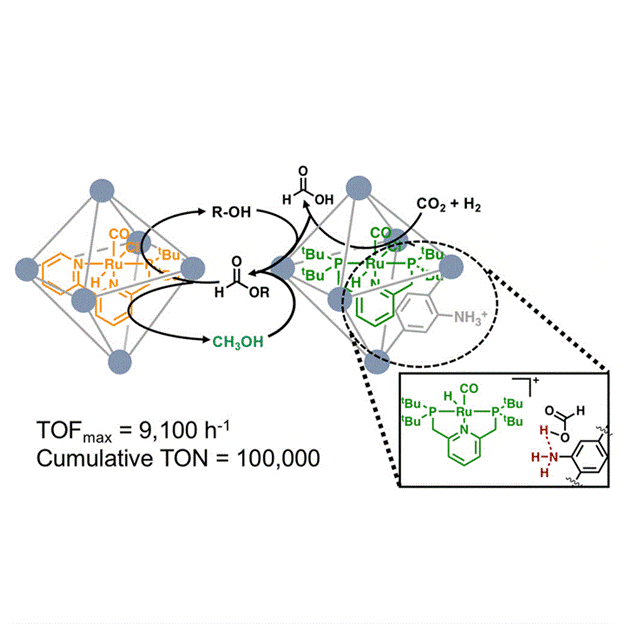
A final area of interest in our laboratory is the use of transition metal catalysts encapsulated in three dimensional matrices for the reversible hydrogenation of carbon dioxide to formic acid and/or methanol. This process would provide a means to store and transport hydrogen, a leading candidate to replace fossil fuels as a sustainable and renewable energy source.
Recent Publications
Apr 7, 2023
Macromolecules
“Adding Polypetides to the Toolbox for Redox-Switchable Polymerization and Copolymerization Catalysis”
Apr 6, 2023
Nature Reviews Methods Primers
“Spatiotemporal control for integrated catalysis”
Nov 4, 2022
Green Chemistry
“N-carboxyanhydrides directly from amino acids and carbon dioxide and their tandem reactions to therapeutic alkaloids”
Oct 12, 2021
Org. Process Res. Dev.
“Air-Stable Iron-Based Precatalysts for Suzuki-Miyaura Cross-Coupling Reactions between Alkyl Halides and Aryl Boronic Esters”